Special reactions of terminal alkynes
Reactions with strong bases
Terminal alkynes treated with strong bases such as sodium amidide in liquid ammonia form relatively stable anions. Such anions are nucleophiles that can be used in substitution reactions.
One of the possible applications is its use on primary or secondary alkyl halides for the preparation of substituted alkynes.
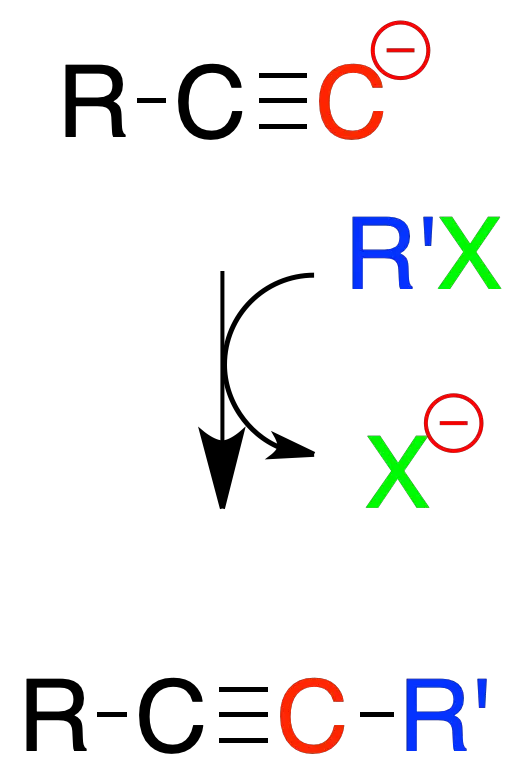
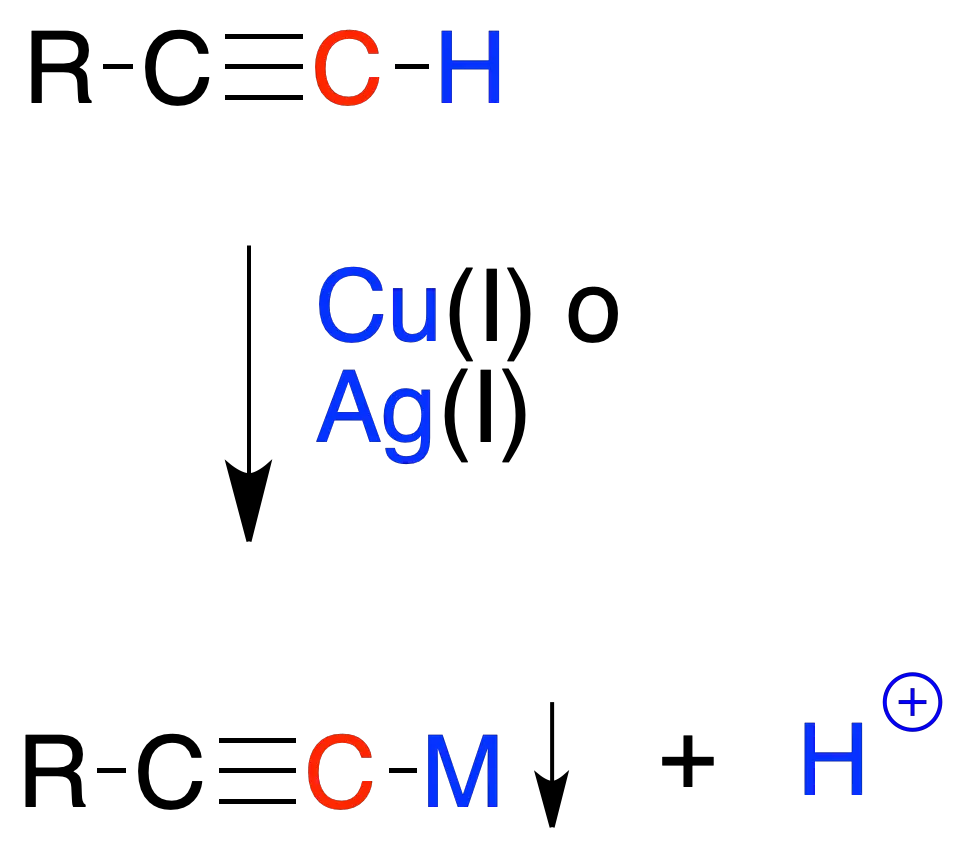
Metallic salts formation
Terminal alkynes form insoluble copper(I) or silver(I) salts, which allows them to be distinguished from disubstituted alkynes (where no such precipitates are formed).
Reduction of alkynes
Catalytic hydrogenation
The catalytic hydrogenation of an alkyne with a metal catalyst (Pd, Pt, Rh, Ni, etc.) leads to the formation of an alkane with the same number of carbons as the starting product.
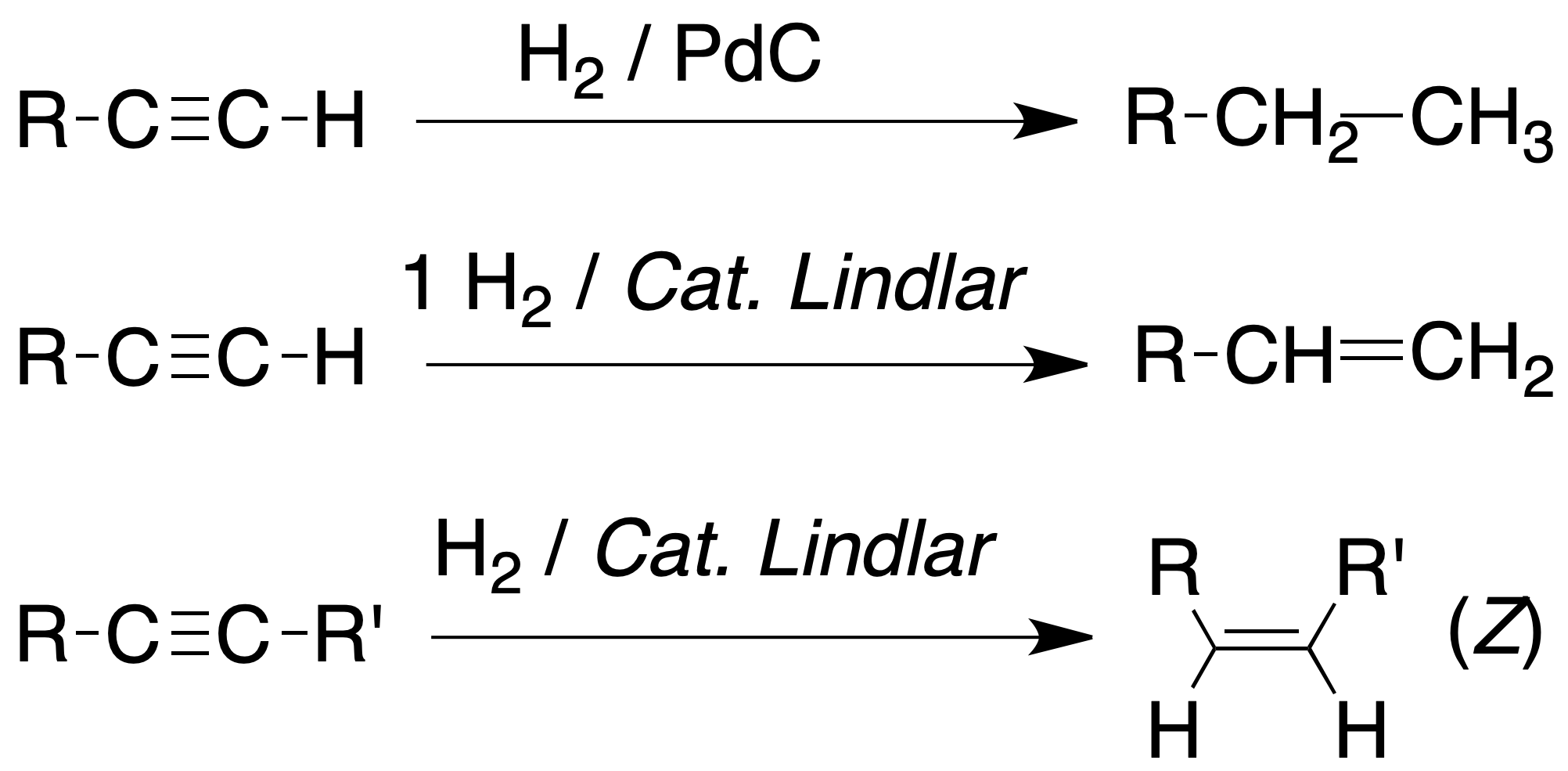
The catalyst is usually a finely divided transition metal and sometimes bonded to the surface of an inert support.
However, using 1 mole of hydrogen, one alkene is produced. Furthermore, if modified catalysts are used, it is possible to control the stereochemistry of the process.
For example, partially deactivated catalysts, such as the Lindlar catalyst (5% Pd / CaCO3 / Pb(AcO)2 / quinoline), produce incomplete hydrogenation of alkynes generating an alkene of Z stereochemistry.
Reduction with metals
Treatment of an alkyne with lithium or sodium metal in liquid ammonia produces alkenes with E stereochemistry via an electron transfer process to the π-system of the alkyne.
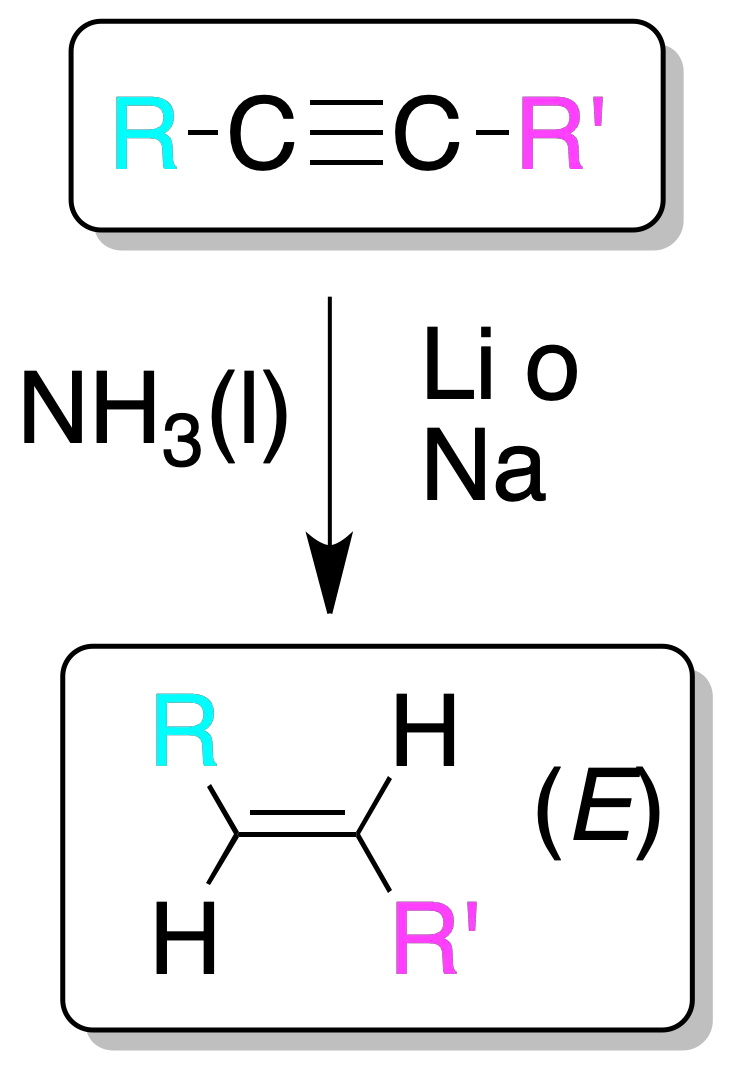
Electrophilic addition of alkynes
Like alkenes, alkynes undergo electrophilic additions.
Acid-catalyzed hydration
Alkynes add a water molecule in the presence of a mineral acid such as H2SO4 and salts of Hg2+. As a result, an enol is obtained which rapidly evolves into the corresponding carbonyl compound.
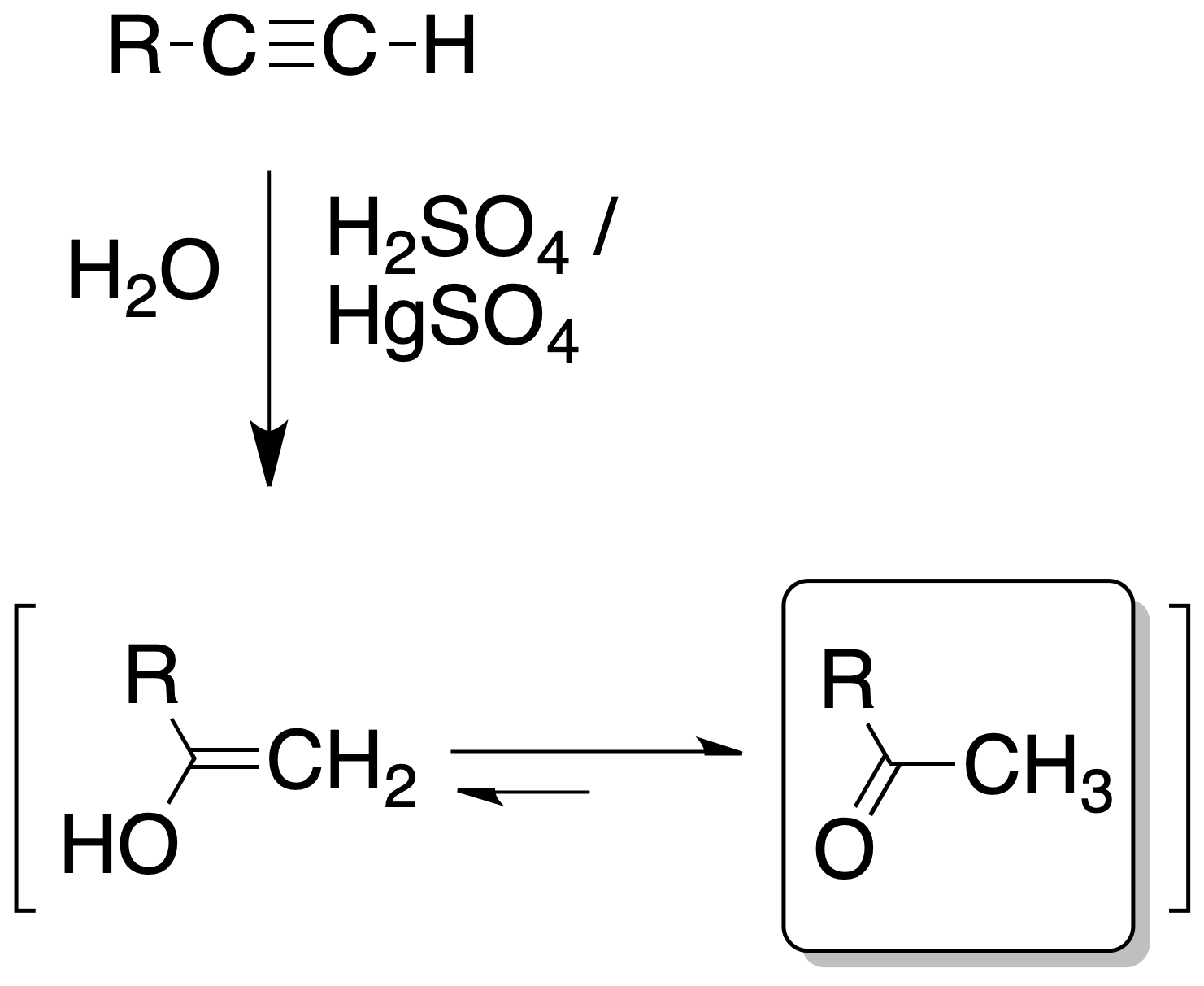
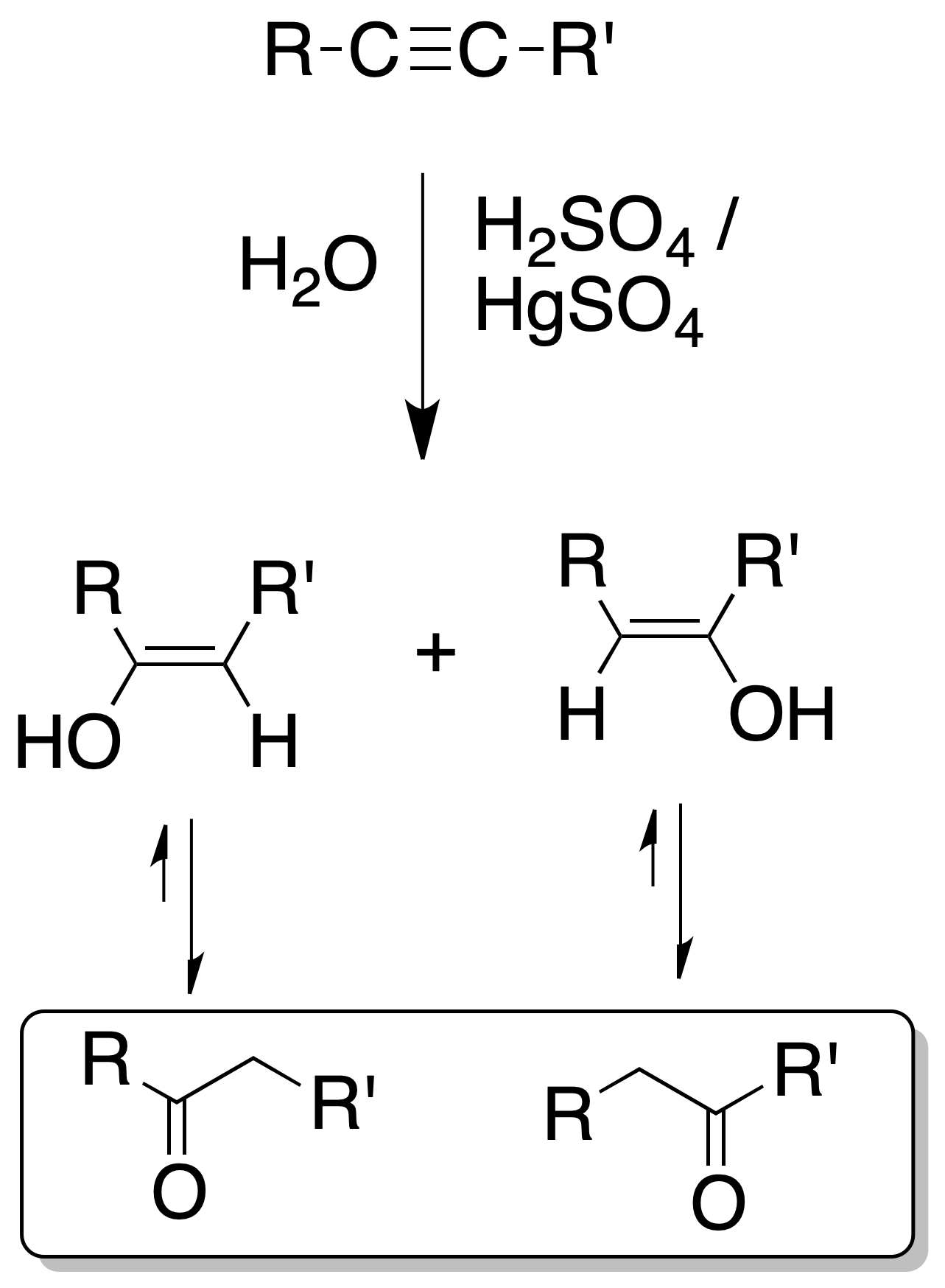
The addition of water is of the Markovnikov type, whereby in monosubstituted alkynes ketones are obtained.
In the case of disubstituted it gives rise to a mixture of ketones.
Hydroboration
The reaction of borane with alkynes leads to the formation of a vinylborane, so that the boron atom is associated with the least substituted carbon (anti-Markovnikov type addition), the steric factor predominating.
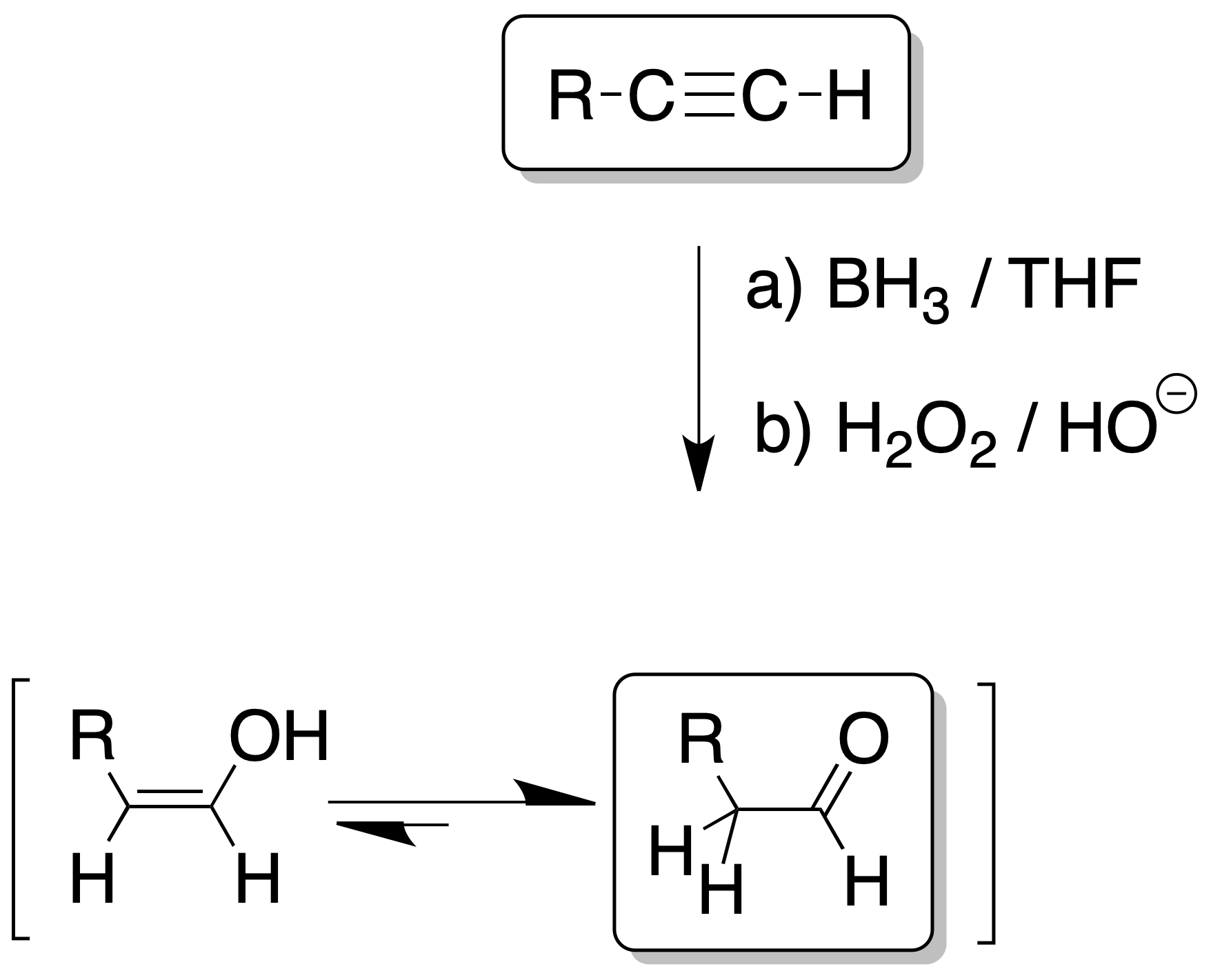
Oxidation with H2O2 of vinylborane produces an enol, which rapidly evolves to the carbonyl compound in a manner similar to the previous reaction.
If the triple bond of the starting alkyne is at the end of a carbon chain, an aldehyde is obtained.
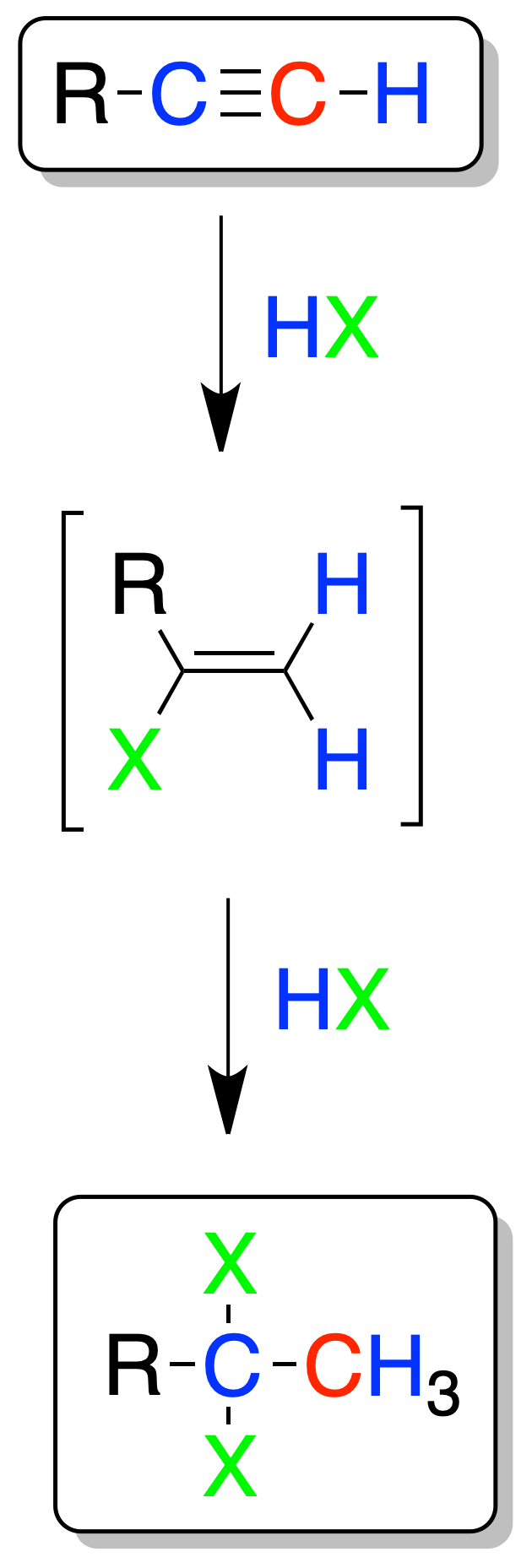
Electrophilic addition of HX
This reaction is carried out sequentially, so that a first molecule is added with formation of a vinyl halide, followed by the addition of a second molecule of HX.
The addition is of the Markovnikov type and leads to the formation of a geminal dihalide.
In the case of terminal alkynes, the halogen atoms are attached to the secondary carbon.
Bromine addition
Alkynes react with bromine to give tetrahalogenated compounds.
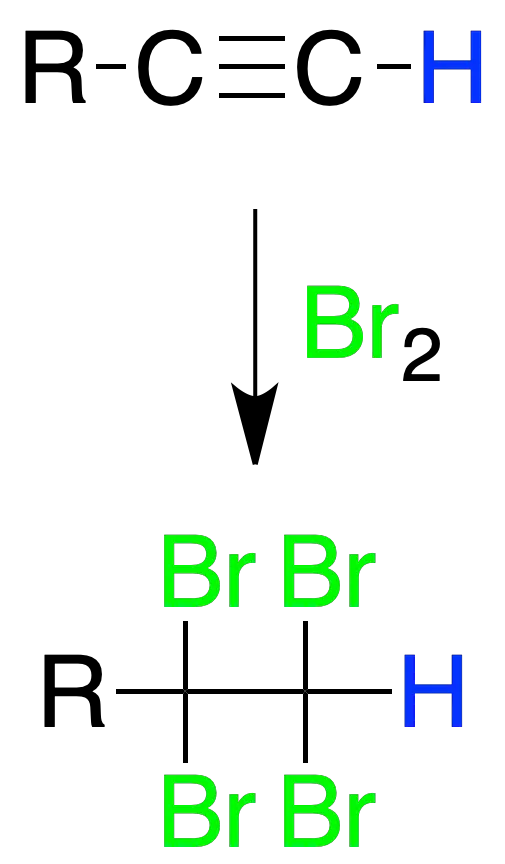
If the reaction is carried out with equimolecular amounts of alkyne and bromine, it is possible to isolate the dibrominated derivative resulting from the addition of 1 mol of bromine.
The reaction is similar to the addition of halogens to double bonds. It occurs via an intermediate bromonium ion and is an anti-type addition.
Radical addition to alkynes
The addition of HBr, to alkynes in the presence of radical initiators takes place with a regiochemistry opposite to electrophilic addition (anti-Markovnikov addition).
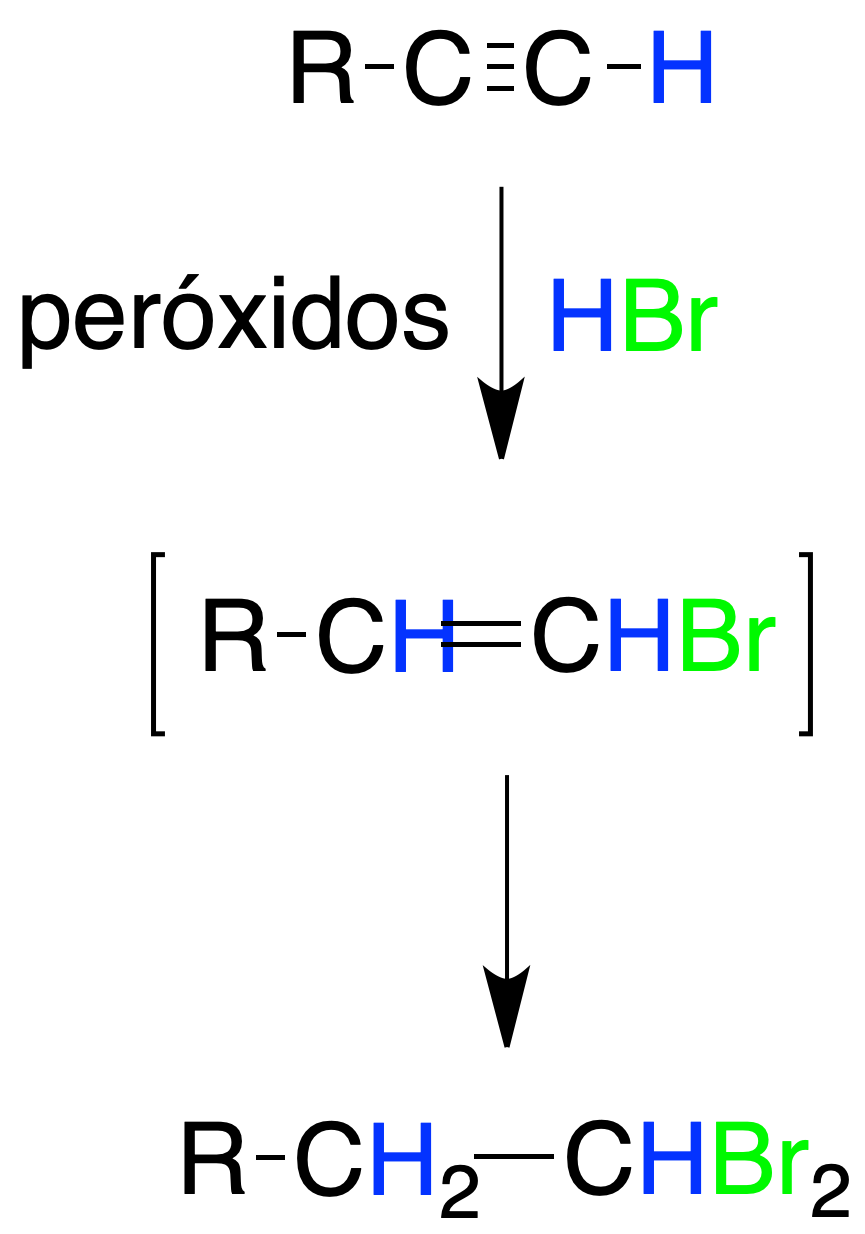
The reaction mechanism is of the radical type, and in some cases it is possible to isolate the vinyl bromide obtained by addition of 1 mol of HBr. If there is an excess of HBr a geminal dihalide is obtained.
Oxidation of alkynes
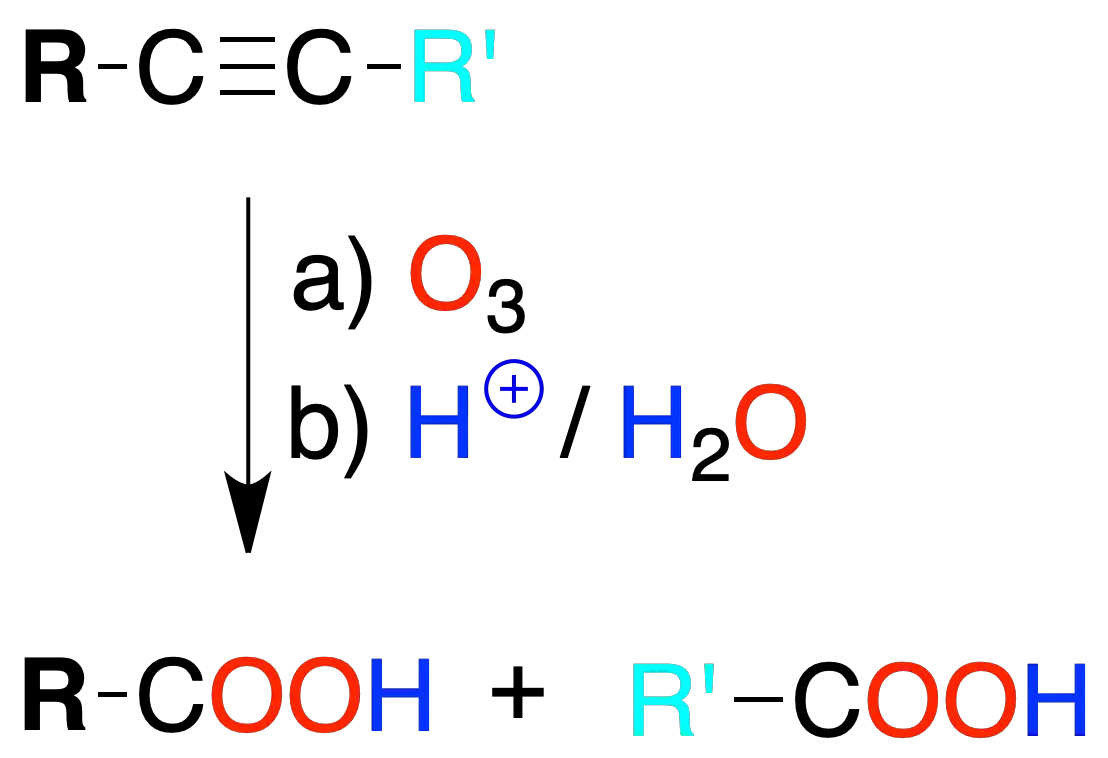
Ozonolysis
The reaction of an alkyne with ozone, followed by treatment with aqueous medium, leads to the formation of carboxylic acids. In the case of a terminal triple bond CO2.
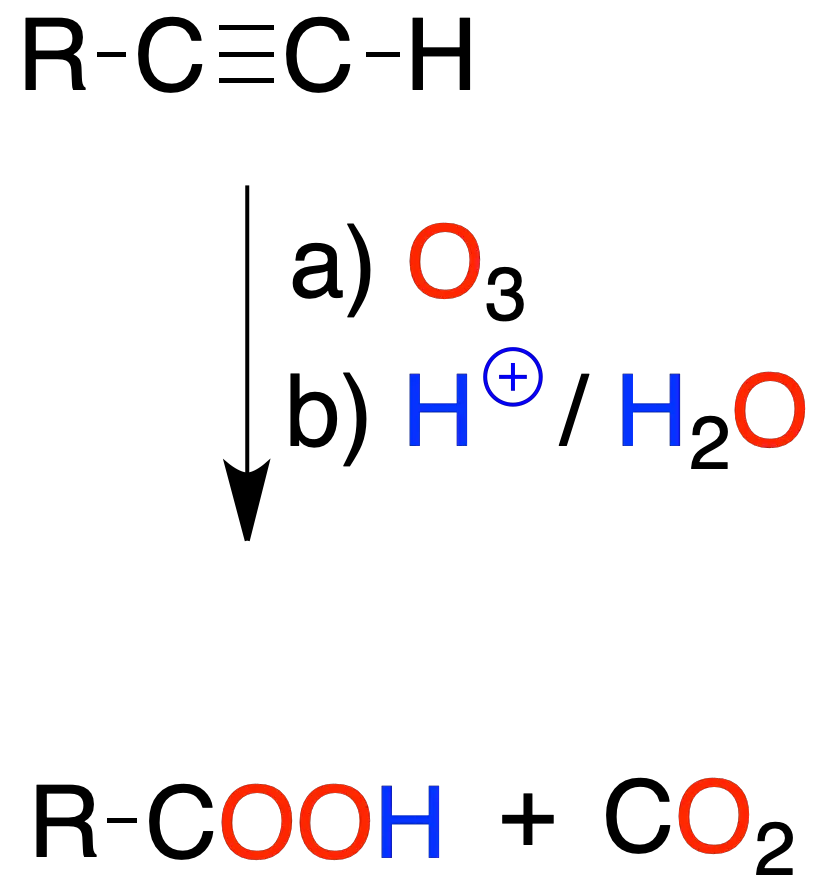
As with alkenes, the reaction is used to characterize the structure of the alkyne by analyzing the fragments obtained.
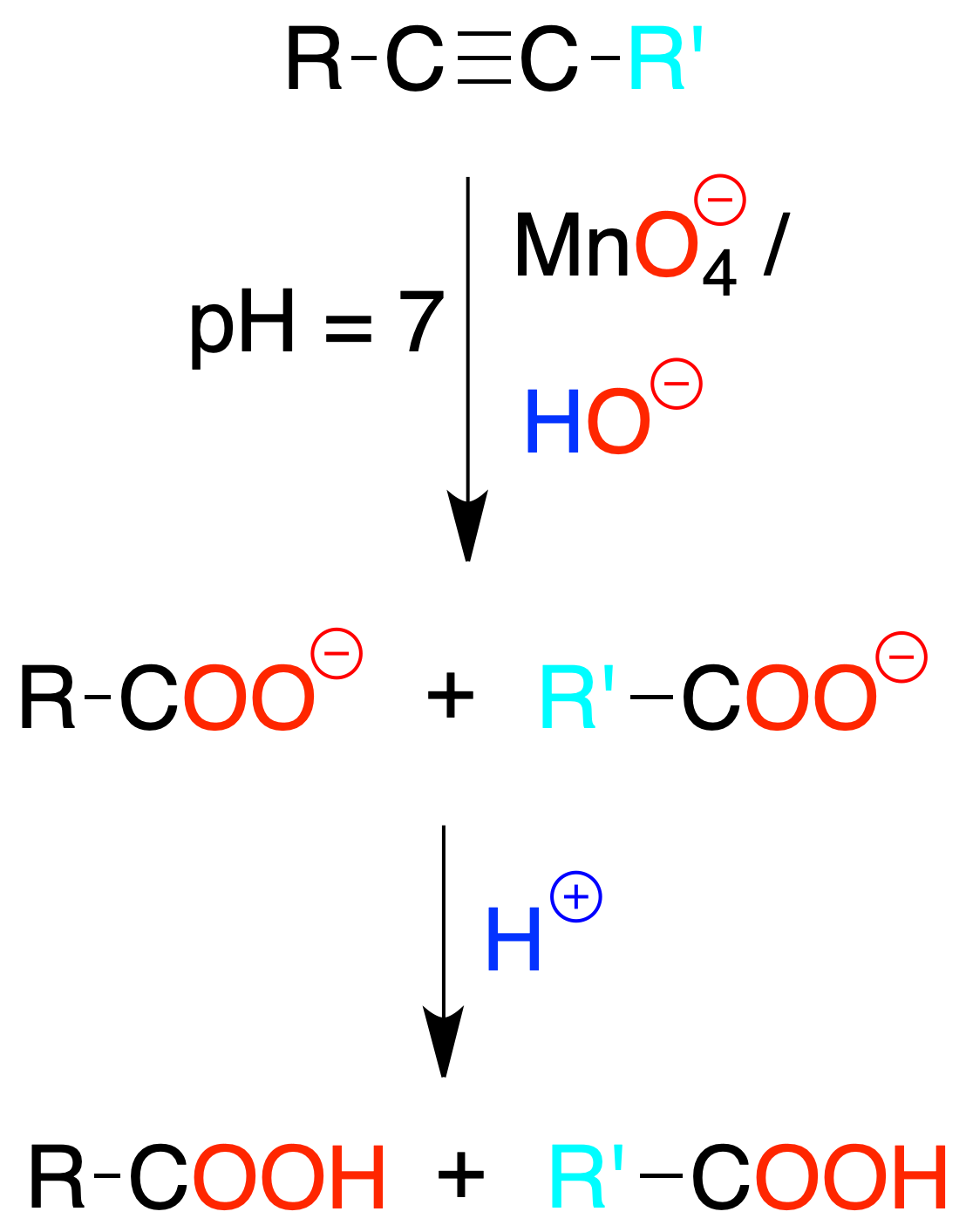
Reaction with permanganate
Potassium permanganate reacts with alkynes in a basic medium producing oxidative cleavage to give the potassium salt of the carboxylic acids. The free acid is obtained by acidifying.
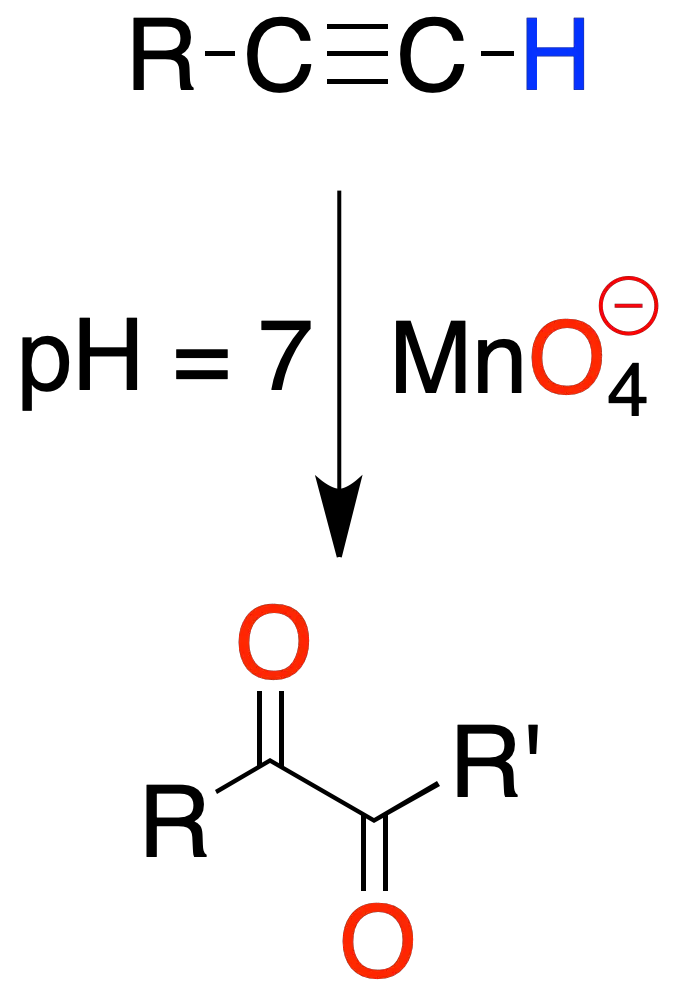
In alkynes disubstituted at neutral pH and cold, a dicarbonyl compound is obtained, with two C=O groups on the carbons presenting the triple bond.
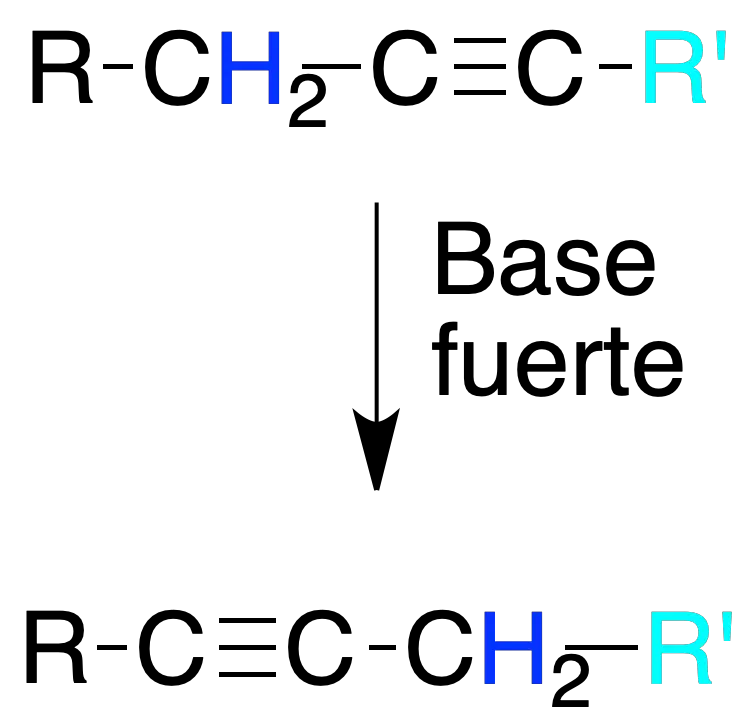
Isomerization of alkynes
Disubstituted alkynes treated with strong bases can give other isomeric alkynes by migration of the triple bonds.