In the analysis of nitrocompounds, one of the characteristics of these compounds is that most of them are colorless liquids.
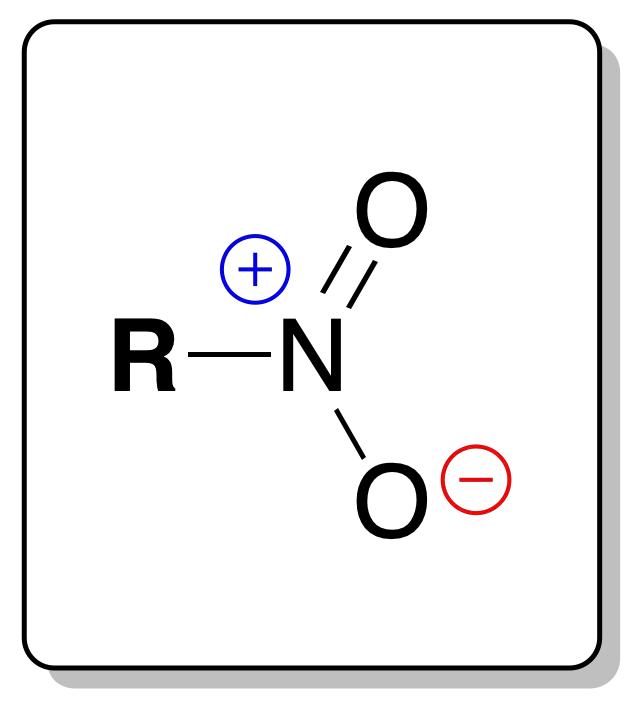
They turn yellow when stored. They have characteristic odors, are insoluble in water and are denser than water. Most aromatic nitrocompounds are yellow crystalline solids.
In the IR spectrum, they show two strong bands at 1560 and 1350 cm-1. Generally, in the analysis of nitrocompounds, their chemical analysis is not used, but they can be reduced to a great variety of nitrogen products.
Zinc test
Procedure : 500 mg of product is dissolved in 10 ml of 50 % EtOH, 500 mg of ammonium chloride and 500 mg of zinc powder are added and the mixture is stirred and heated to boiling point.
Let cool for 5 min, filter and test the Tollens reagent on the filtrate. The unknown product is usually reduced to a hydrazine, hydroxylamine or an amino phenol, compounds easily oxidized with the Tollens reagent.