
Manganese is a chemical element with the symbol Mn and atomic number 25. Manganese, like magnesium, is named after the Greek region of Magnesia, where there are many manganese ores, including the colorful rodochrosite mineral. The pure form of the metal is obtained mainly from the pyrolusite mineral, through a process called electrolysis.
Pure manganese is dense, hard and brittle. This element is present in seawater in the form of manganese hydroxide and manganese oxide, which have accumulated in layers over millions of years to form masses on the seabed. The human body needs a small amount of manganese, which we can obtain from mussels, nuts, oats and pineapples. Applications of manganese include its use in strengthening steel, which is used in the manufacture of railroad tracks and tank armor. Certain dry cell batteries carry a mixture containing manganese oxide. Manganese compounds are also added to gasoline and are used to clean impurities from glass to lighten it or give it a purple color. In prehistoric times, manganese dioxide, MnO2, was crushed to make the dark colors used in cave paintings.
Manganese is also a component of certain enzymes, and it is also used in the production of fertilizers and other chemicals, and in the manufacturing of ceramics and glass.
Chemical properties
Manganese is an essential element in animals: it increases bone strength, helps absorb vitamin B1 and is an important cofactor for enzymes.
Manganese metal is readily oxidized in the presence of oxygen gas, water, and hydrochloric acid. For example, in hydrochloric acid, the following reaction takes place:
Mn(s) + 2HCl(aq) → MnCl2(aq) + H2(g)
In compounds, manganese can be in any of the oxidation states from “+2” to “+7,” inclusive. Examples of manganese in these states are illus- trated in the following series: Mn «0»; Mn2+ «+2»; Mn3+ «+3»; MnO2 «+4»; MnO3– «+5»; MnO42-«+6»; MnO4– «+7».
As it is in group VIIA, its “+7” oxidation state is the preferred one.
The manganic ion (Mn3+) tends to be unstable in aqueous solution and disproportionates into Mn(II) and Mn(IV), as shown in the following reaction:
2Mn3+(aq) + 2H2O(l) → Mn2+(aq) + MnO2(s) + 4H+(aq)
Disproportionation means that an element initially in only one oxidation state changes to two products: one product exhibiting a lower oxidation state and the other exhibiting a higher state.
The oxides formed in the “+2” and “+3” states (manganous oxide (MnO) and manganic oxide (Mn2O3), respectively) are bases due to their solubility in acid solutions, as shown in the following example:
MnO(s) + 2HCl(aq) → MnCl2(aq) + H2O(l)
In the “+4” state, manganese forms black manganese dioxide (MnO2), which is the only compound of manganese (IV). Manganese dioxide is amphoteric because it is soluble in both acid and basic solutions. In higher oxidation states, oxides of manganese are acidic because they dissolve in basic solutions.
The manganate ion (MnO42-) disproportionates in acidic solution to the “+4” and “+7” states, as shown in the following reaction:
3MnO42-(aq) + 4H+(aq) → MnO2(s) + 2MnO4– (aq) + 2H2O(l)
where MnO4– is the purple-colored permanganate ion in which manga- nese is in the “+7” oxidation state.
In acidic solution, permanganate also oxidizes the manganous ion (the “+2” state) to form manganese dioxide (the “+4” state), as shown in the following equation:
2MnO4–(aq) + 3Mn2+(aq) + 2H2O(l) → 5MnO2(s) + 4H+(aq)
In this reaction, manganese begins in two different oxidation states and forms a product that exhibits an oxidation state intermediate between the two initial states.
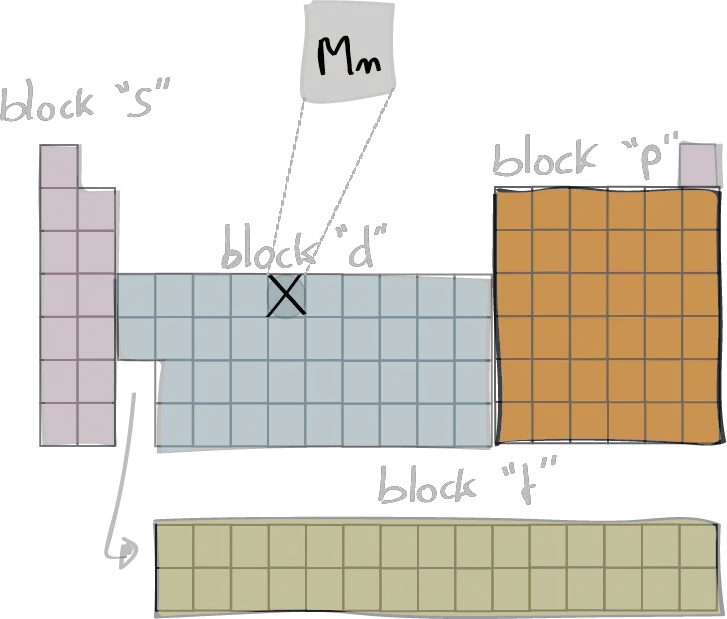
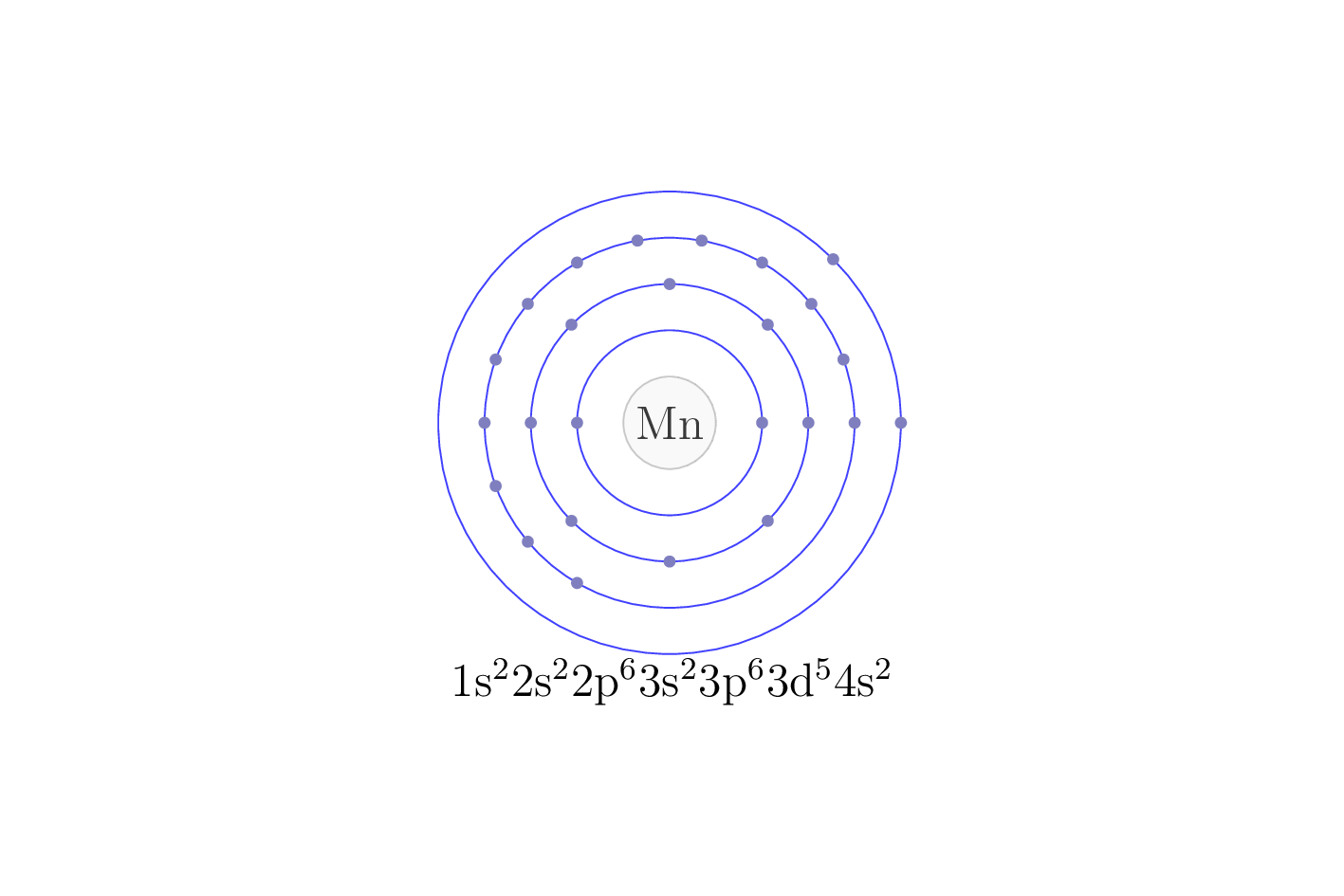
Electron configuration
The electron configuration of an element describes the arrangement of electrons in the atoms of that element, and be used to predict its chemical properties and reactivity.
In the electron configuration notation, the letters "s", "p", "d", and "f" represent the different types of atomic orbitals, and the superscripts indicate the number of electrons in each orbital. The orbitals are filled in a specific order, starting with the lowest energy orbital and working up.
Emission spectra
Each element in the periodic table presents its own unique emission spectra, which is determined by the energy levels of its electrons. When an electron in an atom is excited to a higher energy level, it can de-excite by emitting a photon of light with an energy equal to the difference between the two levels. This results in a characteristic emission line in the spectra (which corresponds to specific wavelengths of light). These spectra are usefull to identify the elements present in a sample.

Symmary of properties (Mn)
| Atomic weight | 54.938043(2) |
| Discoverer (year) | Gahn, Johan Gottlieb (1774) |
| Natural form | metallic solid (cubic centred) |
| Electron configuration | [Ar] 3d5 4s2 |
| M.p. (ºC) | 1245 |
| B.p. (ºC) | 1962 |
| Earth's crust abundance (ppm) | 950 |
| Isotope (abundance %) | 55Mn (100) |
| Density (g/cm3) | 7.43 |
| vdW radius (pm) | 205 |
| Covalent radius (pm) | 129 |
| Electronegativity (Pauling) | 1.83 |
| Vaporisation enthalpy (Kj/mol) | 219.70 |
| Fusion enthalpy (kJ/mol) | 12.91 |
| Specific heat capacity (J/g·K) at 25ºC and 1 at | 0.48 |
| Thermal conductivity (W/cm·K) at 25 ºC and 1 at | 0.080 |
| Oxidation number | +7, +4, +3, +2 |
| Electronic affinity (eV) | unstable ion |
| 1st Ionization energy (eV) | 7.4340 |
Definition of terms in the previous table
- Atomic weight: The average mass of an element's atoms, typically given in atomic mass units (amu).
- Natural form: The most stable and abundant form of an element that occurs naturally in the environment.
- Electron configuration: The arrangement of electrons in an atom or molecule.
- Melting point: The temperature at which a solid substance turns into a liquid.
- Boiling point: The temperature at which a liquid substance turns into a gas.
- Earth's crust abundance (ppm): The concentration of an element in the Earth's crust, typically given in parts per million (ppm).
- Isotope (abundance %): A variant of an element that has the same number of protons in the nucleus, but a different number of neutrons. The abundance of an isotope is the percentage of the isotope in a sample of the element.
- Density (g/cm3): The mass of a substance per unit volume.
- vdW radius (pm): The radius of an atom or molecule as predicted by the van der Waals model, typically given in picometers (pm).
- Covalent radius (pm): The distance from the center of an atom to the center of another atom with which it is bonded covalently, typically given in picometers (pm).
- Electronegativity (Pauling): A measure of an atom's ability to attract electrons in a chemical bond, based on the Pauling scale.
- Vaporisation enthalpy (kJ/mol): The amount of energy required to convert a substance from a liquid to a gas at a constant temperature.
- Fusion enthalpy (kJ/mol): The amount of energy required to convert a substance from a solid to a liquid at a constant temperature.
- Specific heat capacity (J/g·K) at 25ºC and 1 at: The amount of heat required to raise the temperature of 1 gram of a substance by 1 degree Celsius at a constant pressure.
- Thermal conductivity (W/cm·K) at 25 ºC and 1 at: The ability of a substance to conduct heat, typically given in watts per centimeter per kelvin.
- Oxidation number: A positive or negative integer that represents the number of electrons that an atom has gained or lost in a chemical compound.
- Electronic affinity: The energy change associated with adding an electron to a neutral atom to form a negative ion.
- 1st Ionization energy: The energy required to remove the most loosely bound electron from a neutral atom.