Objective
Learn how different organic compounds can be isolated according to their acid-base properties, which change their solubility in organic and aqueous solvents, by liquid-liquid extraction.
 |  |
| 3D structure | 3D structure |
 |  |
| 3D structure | 3D structure |
Background
Liquid-liquid extraction is one of the most common basic operations in the organic chemistry laboratory, since many reactions involve the use of this technique for product isolation.
Carboxylic acids and phenols can react with bases such as sodium bicarbonate, sodium carbonate or sodium hydroxide, producing a proton and forming the corresponding water-soluble anions. In addition, amines produce water-soluble ions generated by protonation, such as ammonium salts.
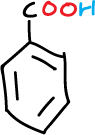
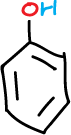
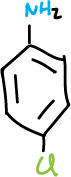
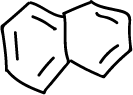
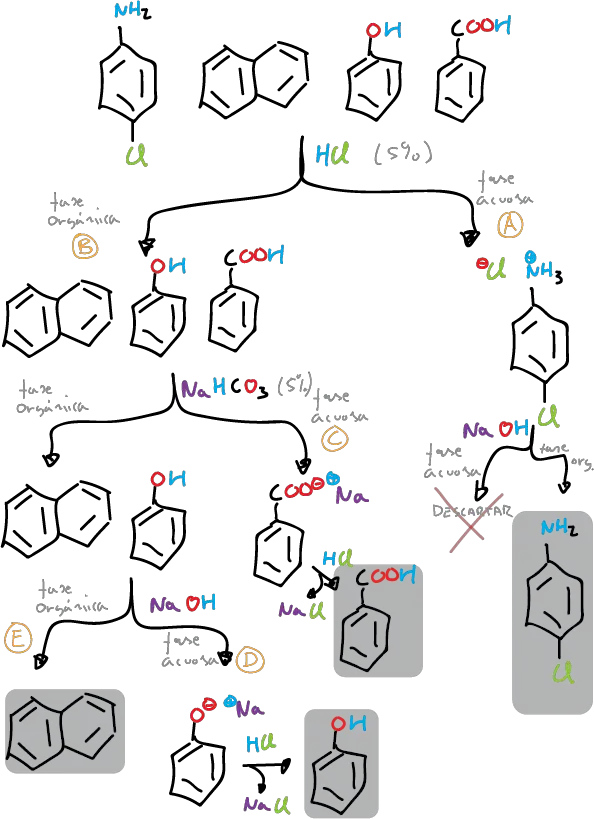
This experiment consists in the separation of the components of a mixture formed by naphthalene, p-chloroaniline, phenol and benzoic acid (5 g/L for each component), which will be dissolved in an organic solvent such as methylene chloride (CH2Cl2), depending on the acidic or basic character of the components of the mixture.
Procedure
Separation of four components (mixture of an acidic, basic and neutral compound):
Take 30 ml of CH2Cl2 solution containing four different dissolved compounds with neutral, basic and acidic character, such as naphthalene, p-chloroaniline, benzoic acid and phenol, respectively.
The amount of each in the 30 ml solution is approximately 4 ml (or 4 g, if solid). Place the solution in a separating funnel and extract with 5 % HCl (2 x 20 ml).
This extraction removes the p–chloroaniline from the organic phase as the water-soluble p-chloroanilinium chloride salt (label flask 1-A), leaving the other three components in organic solution. The aqueous HCl extracts (flask 1-A) are made basic by adding small portions of 10% NaOH, cooling the mixture to a.t. if necessary. Extract this basic solution with CH2Cl2 (2 x 10 ml).
Collect both extracts, dry with anhydrous Na2SO4, filter and remove the solvent with a rotary evaporator, the product in this case being the basic component of the mixture, p-chloroaniline.
Subsequently, extract the organic layer with a 5 % aqueous NaHCO3 solution (2 x 20 ml) (label flask 2-A). This removes the benzoic acid from the organic solution (1-O) as sodium benzoate, but leaves the less acidic phenol and naphthalene in the organic layer (1-O). Acidify the basic aqueous extracts (from flask 2-A) by adding small portions of HCl (conc.). Cool the mixture (flask 2-A) and filter the crystalline product (benzoic acid) under vacuum in a Büchner.
Check that in the mother waters coming from 2-A, adding a small amount of HCl does not cause more precipitate to appear. If so, add a little more acid and filter again in the Büchner itself. Wash the benzoic acid crystals with 20 ml of cold water and dry by letting a stream of air (from the vacuum) through the Büchner.
The remaining organic layer (from flask 1-O) is then extracted with 5% NaOH. The phenol can be extracted from the aqueous layer (flask 3-A) as the water-soluble sodium phenolate salt, leaving the neutral naphthalene as the only compound in the remaining organic layer (flask 2-O).
In this way, each of the 4 components can now be isolated.
Acidify the basic aqueous extracts (flask 3-A) by adding small portions of HCl (conc.) until slightly acidic. Extract this acid solution with CH2Cl2 (2 x 10 ml); after collecting both extracts, dry with anhydrous Na2SO4. Filter and then remove the solvent with a rotary evaporator, giving, in this case, phenol as a liquid compound.
Finally, pour the remaining organic layer (2-O flask) through the separating funnel into an Erlenmeyer flask and dry with anhydrous Na2SO4 for a few minutes. Gravity filter the solution and transfer the filtrate to a rotary evaporator. Naphthalene will appear after removal of the solvent.
General scheme of separation
Physico-chemical properties
This table collects data for the molecular weight (Mw), melting point (M.p.) boiling point (B.p.) and density of the reactives and compounds used in this laboratory experiment.
| Name | Mw (g/mol) | M.p. (ºC) | B.p. (ºC) | Density (g/ml) |
| Benzoic acid | 122.12 | 125 | 249 | 1.08 |
| Naphthalene | 128.17 | 79.5-81.0 | 218 | - |
| NaHCO3 | 84.01 | 300 | - | 2.160 |
| p-Chloroaniline | 127.57 | 72.5 | 232 | 1.140 |
| CH2Cl2 | 84.93 | -97 | 40.0 | 1.33 |
| NaOH | 40.00 | 318 | 1,390 | 2.130 |
| HCl | 36.46 | -30 | >100 | 1.200 |
| Na2SO4 | 142.04 | 884 | - | 2.630 |
| Phenol | 94.11 | 40-42 | 182 | 1.07 |
GHS pictograms
Hazard pictograms form part of the international Globally Harmonized System of Classification and Labelling of Chemicals (GHS) and are collected in the followinf Table for the chemical compounds used in this experiment.
| Name | GHS |
| Benzoic acid |   |
| Naphthalene |    |
| NaHCO3 | Non-hazardous |
| p-Chloroaniline |    |
| CH2Cl2 |  |
| NaOH |  |
| HCl |   |
| Na2SO4 | Non-hazardous |
| Phenol |    |
International Chemical Identifier
The IUPAC InChI key identifiers for the main compounds used in this experiment are provided to facilitate the nomenclature and formulation of chemical compounds and the search for information on the Internet for these compounds.
| Benzoic acid | WPYMKLBDIGXBTP-UHFFFAOYSA-N |
| Naphthalene | UFWIBTONFRDIAS-UHFFFAOYSA-N |
| NaHCO3 | UIIMBOGNXHQVGW-UHFFFAOYSA-M |
| p-Chloroaniline | QSNSCYSYFYORTR-UHFFFAOYSA-N |
| CH2Cl2 | YMWUJEATGCHHMB-UHFFFAOYSA-N |
| NaOH | HEMHJVSKTPXQMS-UHFFFAOYSA-M |
| HCl | VEXZGXHMUGYJMC-UHFFFAOYSA-N |
| Na2SO4 | PMZURENOXWZQFD-UHFFFAOYSA-L |
| Phenol | ISWSIDIOOBJBQZ-UHFFFAOYSA-N |
References
- Isac-García, J.; Dobado, J. A.; Calvo-Flores, F. G.; and Martínez-García, H. (2015). Experimental Organic Chemistry Laboratory Manual. Elsevier Science & Technology. ISBN: 978-0-12-803893-2
Back to the Separation of Components of a Mixture experiment page.